Meet YuiQuery Research
Built for clinical researchers who need an end-to-end workflow from data validation to publication-ready outputs
Do You Struggle With…
Common challenges that slow down clinical research
Data Validation
Verifying dataset accuracy is tedious; missing values and inconsistencies often cause downstream issues
Code & Query Generation
Researchers spend excessive time writing and debugging SQL, Python, or R, leaving non-technical users behind
Visualization & Insights
Moving between tools to create charts, run models, and interpret results slows analysis and introduces errors
5x Faster
From raw data to publication-ready results
From Raw Data to Publication — in One Workflow
YuiQuery Research streamlines the entire journey from raw data to publication
By combining automated data validation, intelligent code generation, guided analytics, and integrated visualization, YuiQuery Research reduces errors, eliminates tool-switching, and accelerates insights — while ensuring compliance and collaboration

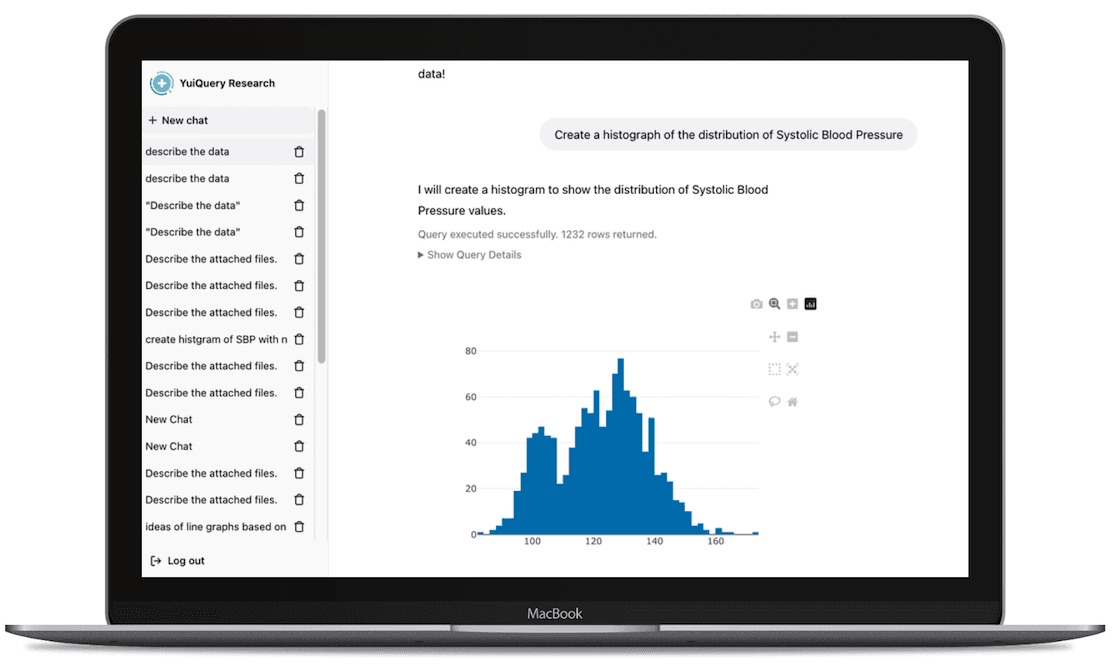
Natural Language Code Generation
Ask questions in natural language and generate validated SQL or Python instantly, expanding access to non-technical users
Guided Analytics
Interpret results with statistical recommendations, plain-language explanations, and transparent assumptions

Visualization Support
Publish findings seamlessly with ready-to-use charts, auto-filled citations, and draft manuscripts from integrated templates
Minimal Metadata Sharing
Protect sensitive information by sharing only anonymized metadata (column names, data types). Your raw data never leaves your environment without explicit approval.
Human-in-the-Loop Approval
Stay in control of PHI by reviewing any real data before it leaves your environment. Every data export requires human approval.
Enforced k-Anonymization
Safeguard patient information by ensuring approved data is shared only in k-anonymized form. Data only appears in groups of 10 or more.
Join the Free Beta
See how YuiQuery Research fits your workflow. Limited spots available.